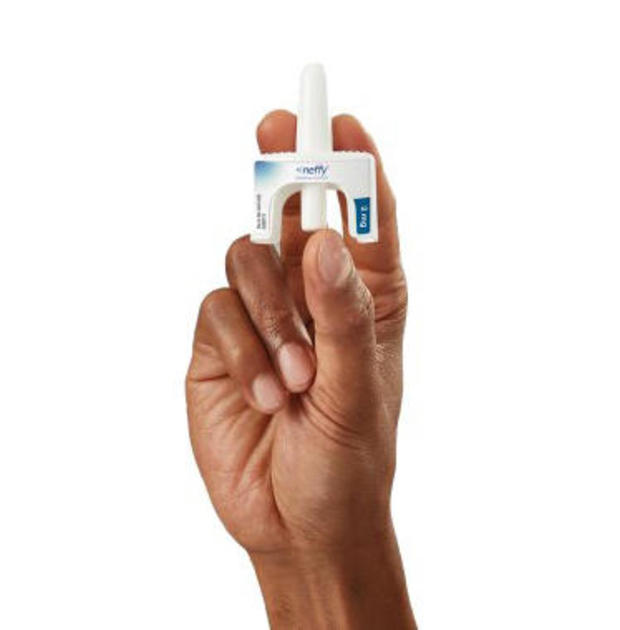
A committee of outside advisers to the Food and Drug Administration voted Thursday in favor of an epinephrine nasal spray product, clearing a key hurdle in what could soon become the first needle-free option for treating severe allergic reactions.
A majority of votes supporting the spray for both adults and children followed a daylong meeting of the agency’s Pulmonary-Allergy Drugs Advisory Committee, which picked up an application from ARS Pharmaceuticals for their proposed epinephrine spray, known as Nefi.
Nefi is designed to deliver a 2 mg dose of epinephrine, which can reverse the symptoms of a life-threatening allergic reaction. The company is seeking FDA approval for anyone weighing at least 30 kilograms — about 66 pounds — and is working on future studies for younger children.
ARS Pharmaceuticals
Consultants wrestled with data gaps raised by the drugmaker’s approach, which was aimed at bypassing large clinical trials instead of comparing them with other previously approved injection products used to treat allergic reactions.
The FDA asked the committee whether it should claim that more study is needed anyway, but acknowledged the steep challenges facing studying life-threatening allergic reactions that already have widely available treatments.
The drugmaker said the FDA is expected to issue a final ruling on whether to approve Nefi “by mid-2023.” If approved, the company told investors in March that it expects to launch the prescription spray by the end of the year.
Although the FDA is not legally bound to follow the advisory committee’s vote, the agency says expert input plays an important role in helping its staff weigh complex questions raised by drug company submissions.
ARS Pharmaceuticals said it hopes Nefi will fill a key unmet need for Americans. Risk of serious allergic reactionsBut it’s uncomfortable using injection alternatives currently on the market — like the EpiPen, first approved by the FDA in 1987
The drugmaker cited a study that found some people could administer its epinephrine spray 18 minutes before the injection. They also say the product is small enough to be easier to carry than its competitors, estimating that 35% more people would have epinephrine with them if the spray form were available.
“If they don’t have it, that’s a moot point. They don’t deliver, they don’t have the drug, they go to the hospital,” Richard Lowenthal of ARS Pharmaceuticals told the committee.
The device used to administer Nefi is already widely used for some other nasal spray drugs on the market, including the overdose reversal drug naloxone — known as Narcan nasal spray — that was recently approved for sale over-the-counter without a prescription.
Why was a meeting necessary?
Instead of conducting extensive clinical trials, drugmakers can often gain approval by showing their products trigger similar measurements in the body than already approved drugs.
In Nefi’s case, ARS Pharmaceuticals collected data on metrics like tracking a person’s blood pressure or the concentration of epinephrine that ended up in their bloodstream.
The company then compared it to other injectable products, with results showing that Nefi was neither significantly superior nor inferior.
But FDA reviewers said they struggled with key uncertainties comparing Nefi’s results with those seen after injections already on the market.
The agency said one of the reasons it needed the committee to discuss was epinephrine’s unique history.
This drug has been marketed to Americans in some form of injection since 1901—before Congress created the FDA—and therefore does not have to go through the modern approval process to prove safety and efficacy.
That leaves little data for the FDA to verify how closely Nefi’s measurements actually translate to efficacy in saving patients. Life-threatening reactions.
More complex comparisons: There was a wide range of measurements collected by company as well as from different injection products. The company’s studies were only from healthy volunteers or mild allergic reactions. Some results have also raised questions about whether this route may be less effective than injections.
“I would hate to learn, without some really good clinical data, that we recommend approval of a product based on surrogate data that is inconsistent and somewhat misleading and ultimately harms patients,” committee member Dr. Louise Nelson said. Lewis Nelson says
But others were skeptical that more research would be possible, or produce results good enough to clear up outstanding questions surrounding the spray’s benefits and risks.
“What more can we reasonably legitimately expect? And if it turns out exactly the way we want it to, do we have that much more confidence than where we’re sitting now? I’m just not sure,” committee member Dr. Leonard Bachrier says
Alexander Tin